Fda approves very first alopecia procedure for problem that brings about sudden hair reduction
People today suffering from a unusual autoimmune disorder that triggers their hair to instantly drop out, generally in clumps, now have a procedure selection for the initial time.
On Monday, the Meals and Drug Administration authorised baricitinib, a once-a-working day capsule created by the drugmaker Eli Lilly to take care of alopecia areata, an autoimmune illness that triggers sudden hair decline. The drug was at first permitted by the Food and drug administration in 2018 to address rheumatoid arthritis.
The drug — component of a course of medications regarded as JAK inhibitors — is the to start with authorised cure in the United States for the ailment, which influences a lot more than 300,000 persons in the region each calendar year, in accordance to the Fda.
People today with the autoimmune condition can experience hair reduction any place on their human body, together with all-around the scalp, eyebrows and eyelashes, in accordance to the American Academy of Dermatology. The issue can develop at any age, even though most men and women acquire it in the course of childhood or for the duration of their teenage yrs, in accordance to the group.
Alopecia recently acquired countrywide notice soon after actor Will Smith infamously slapped Chris Rock at the Oscars after the comic manufactured a joke about actor Jada Pinkett Smith, Smith’s wife, who suffers from a different type of the situation than the 1 taken care of by Eli Lilly’s drug.
Baricitinib helps regrow hair by stopping the body’s immune technique from attacking hair follicles.
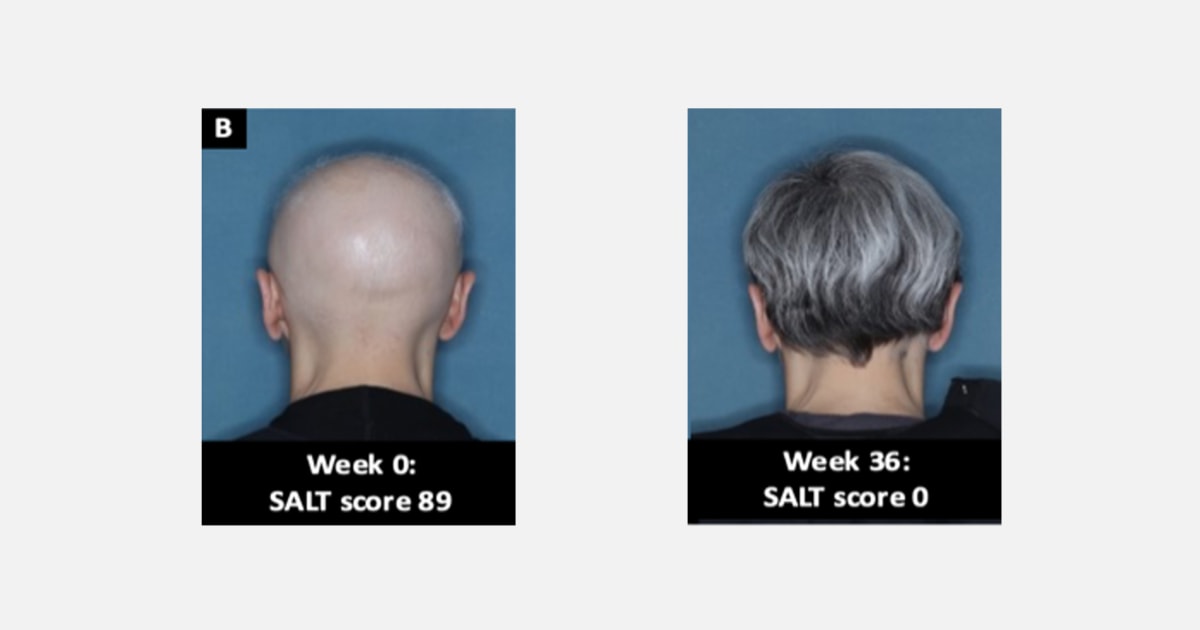
Two section 3 clinical trials, involving a whole of 1,200 individuals with extreme alopecia areata, found that about 40 p.c of people today who obtained a each day 4-milligram dose of the drug regrew all or practically all of their hair after 36 weeks.
That compares to the about 20 p.c of individuals who acquired a decrease 2-milligram dose of the drug and fewer than 6 % of the persons in the placebo team.
People today in the trials experienced dropped at the very least 50 p.c of hair from their scalp prior to having the drug.
The drug is a breakthrough for folks with the autoimmune dysfunction, reported Dr. Brett King, a Yale dermatologist who was the principal investigator for the two trials. Lots of use wigs or scarves to hide their affliction.
“It is really a devastating disorder,” he stated. “Test to visualize you wake up with a spot one day. And then, consider, a few weeks afterwards, or three months later on, or a few several years later, picture all of your hair heading absent.”
Dr. Luis Garza, a dermatologist at the Johns Hopkins College Faculty of Medicine who treats persons with alopecia, explained he’s experienced patients stop their positions or avoid going out in community completely about the tension of dropping their hair. He was not associated in the analysis for the drug.
Up until finally now, there were not lots of possibilities for people with the problem: They often relied on steroid injections or unproven creams in an endeavor to reverse the condition, Garza, who researches hair loss, mentioned.
Men and women can take other drugs that might help, such as JAK inhibitors ruxolitinib or tofacitinib, but they are not accredited by the Fda for alopecia, he claimed. That usually means they can only be approved off-label, which insurers generally really don’t cover and can price sufferers as a great deal as $5,000 a thirty day period, he claimed explained.
The out-of-pocket value for individuals can be “crazy,” Garza mentioned.
The wholesale cost of Eli Lilly’s baricitinib is $2,497.20 for a 30-working day offer of 2-milligram pills, Marlo Scott, a spokesperson for Eli Lilly, explained to G3 Box News News in an email. The business also provides a system that permits people today to fork out $5 or $25 a thirty day period, dependent on no matter whether the affected individual is coated by insurance coverage, she reported.
The most typical facet effects affiliated with the drug contain upper respiratory tract bacterial infections, headache, acne breakouts and high cholesterol, in accordance to the agency.
The medication might not be for every person: Folks with an ongoing infection, cancer or a heritage of coronary heart attack or stroke ought to consult with the medical professional just before thinking about the medication, Garza reported.
King, the principal investigator for the trials, pointed out that two other identical medicine, from Pfizer and Live performance Prescribed drugs, are also in progress to address the ailment and could be approved by the Fda within just the next several decades.
Follow G3 Box News HEALTH on Twitter & Fb.